How to personalize nutrition for functional medicine
Learn how functional medicine practitioners can personalize nutrition using multi-omics data, AI tools, and phased protocols to improve patient outcomes and metabolic health.

Generic nutrition advice rarely works for the complex patients walking into functional medicine clinics. Each person brings a unique genetic blueprint, microbiome composition, hormonal profile, and lifestyle pattern that shapes how they respond to food. Personalized nutrition integrates these multiple data streams to create tailored dietary plans that address root causes and optimize health outcomes. Functional medicine practitioners who master advanced personalization strategies can dramatically improve patient adherence, accelerate healing timelines, and achieve measurable improvements in cardiometabolic markers, gut health, and quality of life. This guide walks you through the essential data collection methods, AI-powered precision tools, phased protocol implementation, and outcome evaluation frameworks you need to deliver truly individualized nutrition care.
Table of Contents
- Key takeaways
- Gathering comprehensive patient data prerequisites
- Integrating multi-omics and AI for precision nutrition strategies
- Implementing phased functional medicine protocols for personalized nutrition
- Evaluating outcomes and addressing limitations in personalized nutrition
- Leverage AI-powered tools to personalize nutrition in your practice
- What types of data are essential for personalizing nutrition?
Key Takeaways
| Point | Details |
|---|---|
| Personalization data fusion | Personalized nutrition combines genetic data, microbiome profiles, hormone status, and lifestyle patterns to tailor dietary plans. |
| Multiomics with AI | Integrating genomics, epigenomics, metabolomics, and microbiome data with AI enables precise, adaptable recommendations. |
| Protocol implementation | A phased protocol of data gathering intervention design and ongoing monitoring supports measurable health improvements. |
| RCT outcomes | Clinical trials show improvements in cardiometabolic markers and mood with personalized nutrition approaches. |
| Practical challenges | Cost, accessibility, and potential overreliance on genetics can limit adoption in routine care. |
Gathering comprehensive patient data prerequisites
Effective personalized nutrition starts with collecting the right data types. You need a complete picture of each patient's biological systems, metabolic function, and daily habits before designing interventions. Tailoring your diet to your unique needs requires integrating genetic profiles, metabolic markers, microbiome composition, hormone levels, and lifestyle patterns into a cohesive assessment framework.
Start with comprehensive lab panels that reveal underlying imbalances. Order standard blood work including lipid panels, fasting glucose, HbA1c, inflammatory markers like CRP, and thyroid function tests. Add methylation biomarkers such as homocysteine, folate, and B12 status to identify genetic variants affecting nutrient metabolism. Gut microbiome sequencing through stool testing provides critical insights into digestive capacity, inflammation, and dysbiosis patterns that shape nutrient absorption and immune function.
Continuous glucose monitors offer real-time metabolic data that static lab values cannot capture. Patients wear CGM devices for 10 to 14 days, generating thousands of glucose readings that reveal postprandial spikes, overnight patterns, and individual food responses. This data helps you identify insulin resistance, reactive hypoglycemia, and specific trigger foods with precision impossible through traditional testing methods.
Patient lifestyle and food-mood tracking adds the behavioral context essential for sustainable change. Have patients log meals, symptoms, energy levels, sleep quality, and stress through apps or journals for at least two weeks before your initial consultation. This qualitative data reveals patterns linking specific foods to symptoms, identifies emotional eating triggers, and establishes baseline adherence capacity.
| Data Type | Tools | Clinical Role |
|---|---|---|
| Genetic profile | SNP panels, methylation tests | Identify nutrient metabolism variants, detox capacity, food sensitivities |
| Metabolic markers | Blood panels, HbA1c, lipids | Assess cardiometabolic risk, insulin resistance, inflammation |
| Microbiome composition | Stool sequencing, SIBO breath tests | Evaluate gut health, dysbiosis, digestive capacity |
| Hormone levels | Salivary cortisol, sex hormones, thyroid | Understand stress response, reproductive health, metabolism |
| Real-time glucose | CGM devices | Capture individual food responses, metabolic flexibility |
| Lifestyle patterns | Food-mood journals, wearables | Track adherence, symptom triggers, behavioral context |

Pro Tip: Start with functional nutrition checklists to ensure you collect all essential data points systematically before designing personalized plans. Missing even one data category can lead to incomplete assessments and suboptimal interventions.
Integrate findings from multiple sources rather than relying on single test results. A genetic variant affecting folate metabolism matters more when combined with low serum folate and elevated homocysteine. Microbiome dysbiosis gains clinical significance when paired with food-mood logs showing bloating after specific meals. This multi-modal approach, detailed in functional medicine nutrition protocols, creates the foundation for truly personalized interventions.
Integrating multi-omics and AI for precision nutrition strategies
Once you have comprehensive patient data, the challenge becomes synthesizing complex information into actionable recommendations. Multi-omics integration combines genomics, epigenomics, metabolomics, and microbiome data to predict optimal nutrition strategies with unprecedented precision. Personalized nutrition and multiomics analyses demonstrate how AI and machine learning algorithms process these datasets to improve weight management, glycemic control, and metabolic health markers in trials like PREDICT and FOOD4ME.
AI-powered platforms analyze thousands of data points simultaneously, identifying patterns invisible to manual review. Machine learning models trained on large population datasets can predict individual responses to specific foods based on genetic variants, microbiome composition, and metabolic markers. These algorithms generate personalized macronutrient ratios, meal timing recommendations, and food lists tailored to each patient's unique biology.

The PREDICT study tracked over 1,000 participants wearing CGMs while consuming standardized meals, revealing massive individual variation in glucose and lipid responses to identical foods. Some patients showed glucose spikes after white bread but stable responses to ice cream, while others demonstrated opposite patterns. AI algorithms identified the combination of genetic, microbiome, and metabolic factors predicting these responses, enabling truly personalized dietary guidance.
FOOD4ME trial results showed participants receiving AI-powered nutrition plans achieved significantly greater improvements in dietary quality, nutrient intake adequacy, and cardiometabolic markers compared to generic advice groups. The precision enabled by multi-omics integration translated into measurable clinical benefits that persisted at six-month follow-up.
| Approach | Data Sources | Personalization Level | Clinical Outcomes |
|---|---|---|---|
| Traditional nutrition | Food frequency questionnaires, basic labs | Low (population-based guidelines) | Modest improvements, high variability |
| Genetic-only personalization | SNP panels | Medium (single-omics) | Small effect sizes, limited predictive power |
| AI-enabled multi-omics | Genomics, metabolomics, microbiome, CGM, lifestyle | High (individual prediction models) | Significant improvements in weight, glucose, lipids, adherence |
Pro Tip: Always validate AI-generated recommendations against clinical judgment and patient-specific factors. Algorithms excel at pattern recognition but cannot account for social determinants, food access limitations, cultural preferences, or psychological barriers that shape real-world adherence. Use AI in functional medicine personalized care tools as decision support, not replacement for clinical expertise.
Implement AI tools that integrate seamlessly with your existing workflow rather than creating parallel systems. Look for platforms that pull data directly from lab interfaces, sync with patient tracking apps, and generate recommendations in formats you can easily customize. The goal is augmenting your clinical intelligence, not adding administrative burden.
Implementing phased functional medicine protocols for personalized nutrition
Translating data and AI insights into patient care requires structured protocols that guide assessment, intervention, and monitoring phases. Nutrition and functional medicine protocols emphasize systems biology approaches that address gut health, detoxification pathways, and hormonal balance as interconnected systems rather than isolated issues.
-
Conduct comprehensive initial assessment gathering all data types outlined previously, including genetic testing, metabolic panels, microbiome analysis, hormone evaluation, and lifestyle tracking. Schedule 60 to 90 minutes for this appointment to review findings thoroughly and establish baseline measurements.
-
Identify primary dysfunctional systems driving symptoms using assessment data. Common patterns include gut dysbiosis with leaky barrier, impaired methylation affecting detoxification, hormonal imbalances disrupting metabolism, or mitochondrial dysfunction limiting energy production.
-
Design phased interventions starting with foundational support before adding targeted therapies. Begin with elimination diets removing common triggers like gluten, dairy, and processed foods for 4 to 6 weeks while supporting gut healing with bone broth, fermented foods, and anti-inflammatory protocols.
-
Layer in personalized nutraceuticals based on genetic variants and lab findings. Patients with MTHFR variants need methylated B vitamins, those with low vitamin D require higher dose supplementation, and individuals with oxidative stress benefit from targeted antioxidants like glutathione or NAC.
-
Adjust macronutrient ratios and meal timing according to metabolic type and CGM data. Insulin-resistant patients often thrive on lower carbohydrate approaches with intermittent fasting windows, while others need balanced macros with frequent meals to stabilize blood sugar.
-
Implement lifestyle modifications addressing sleep, stress management, and movement patterns that influence nutrient metabolism. Poor sleep disrupts glucose regulation and increases inflammation regardless of dietary quality, while chronic stress depletes nutrients and impairs digestion.
-
Schedule follow-up visits every 4 to 6 weeks during intervention phase to monitor progress, adjust protocols, and maintain accountability. Use repeat labs at 3 to 6 months to measure objective improvements in inflammatory markers, metabolic function, and nutrient status.
Food as medicine represents the foundation of functional medicine, addressing root causes of chronic conditions like autoimmunity, SIBO, and metabolic dysfunction through targeted dietary interventions rather than symptom suppression.
Common therapeutic considerations include:
- Elimination diets for identifying food sensitivities and reducing systemic inflammation
- Low FODMAP protocols for SIBO and IBS management with gradual reintroduction
- Ketogenic or low-carb approaches for insulin resistance and neurological conditions
- Anti-inflammatory Mediterranean-style patterns for cardiovascular and autoimmune support
- Specific carbohydrate diet for inflammatory bowel conditions
- Autoimmune protocol (AIP) for Hashimoto's, rheumatoid arthritis, and other autoimmune presentations
Red flags requiring protocol adjustment include worsening symptoms during elimination phases, inability to tolerate recommended supplements, lack of improvement after 8 to 12 weeks of adherence, or development of new symptoms suggesting underlying issues like functional B12 deficiency despite normal serum levels. These scenarios demand deeper investigation and protocol modification.
Use functional nutrition checklists at each phase to ensure comprehensive coverage of assessment domains, intervention components, and monitoring metrics without missing critical elements.
Evaluating outcomes and addressing limitations in personalized nutrition
Measuring success requires tracking both objective clinical markers and subjective patient-reported outcomes. Focus on metrics that reflect underlying physiology rather than vanity measures alone. Effects of personalized nutrition on cardiometabolic health show patients receiving individualized plans achieve significant reductions in triglycerides, improvements in HbA1c and weight, enhanced microbiome diversity, and are twice as likely to report better mood and satiety compared to generic advice groups.
| Outcome Measure | Expected Improvement | Timeframe | Benchmark |
|---|---|---|---|
| Fasting triglycerides | 15-25% reduction | 3-6 months | Below 100 mg/dL optimal |
| HbA1c | 0.3-0.7% decrease | 3-4 months | Below 5.7% non-diabetic |
| Body weight | 5-10% reduction | 3-6 months | Individualized based on composition |
| Microbiome diversity | 20-30% increase | 3-6 months | Shannon index above 3.5 |
| CRP inflammation | 30-50% reduction | 2-4 months | Below 1.0 mg/L optimal |
| Mood/satiety scores | 40-60% improvement | 6-12 weeks | Patient-reported validated scales |
Track symptoms weekly using standardized questionnaires that capture energy levels, digestive function, sleep quality, mood stability, and pain levels. These subjective measures often improve before lab markers change, providing early validation that interventions are working and maintaining patient motivation.
Recognize significant limitations shaping real-world implementation. Challenges in personalized nutrition adoption include high costs for comprehensive testing, accessibility barriers limiting patient reach, and overhyped genetics contributing small effect sizes compared to behavior change. Cost-effectiveness analysis shows genetic testing alone rarely justifies expense without integration into comprehensive protocols addressing lifestyle factors.
Equity concerns arise when advanced testing and AI tools remain available only to affluent patients with discretionary healthcare spending. Consider tiered approaches offering basic personalization through affordable options like food-mood tracking and CGM trials before layering expensive genetic or microbiome testing for patients who can afford comprehensive workups.
Address genetic determinism misconceptions by emphasizing epigenetics and lifestyle factors that modify gene expression. Patients with APOE4 variants face elevated Alzheimer's risk, but Mediterranean diet adherence, exercise, and stress management significantly reduce that risk regardless of genetics. Frame genetic insights as informing strategy rather than determining destiny.
Best practices for maximizing effectiveness:
- Start with highest-yield, lowest-cost interventions before adding expensive testing
- Prioritize behavior change support and accountability over perfect data collection
- Use AI tools to enhance efficiency, not replace therapeutic relationship
- Validate all recommendations against patient values, preferences, and practical constraints
- Monitor outcomes systematically using both objective markers and patient-reported measures
- Adjust protocols based on individual response rather than rigid adherence to initial plans
- Address social determinants and food access barriers that undermine even optimal recommendations
Common pitfalls include over-testing without clear clinical utility, paralysis by analysis delaying intervention, rigid protocol adherence despite poor patient response, and neglecting psychological factors driving eating behaviors. The most sophisticated personalization strategy fails without addressing motivation, self-efficacy, and environmental barriers to implementation.
Leverage AI in functional medicine personalized care platforms that automate data synthesis and recommendation generation, freeing your time for the high-touch coaching and troubleshooting that drives sustainable behavior change. Technology should enhance rather than replace the therapeutic alliance central to functional medicine success.
Leverage AI-powered tools to personalize nutrition in your practice
Implementing the comprehensive personalization strategies outlined above requires significant time investment that most practitioners cannot sustain manually. Meelio's AI-powered platform automates data integration, personalized nutrition planning, and care plan generation specifically for functional medicine protocols. Our system synthesizes genetic results, lab panels, microbiome reports, and patient tracking data into actionable recommendations aligned with frameworks like Bredesen, Wahls, and Shoemaker protocols.
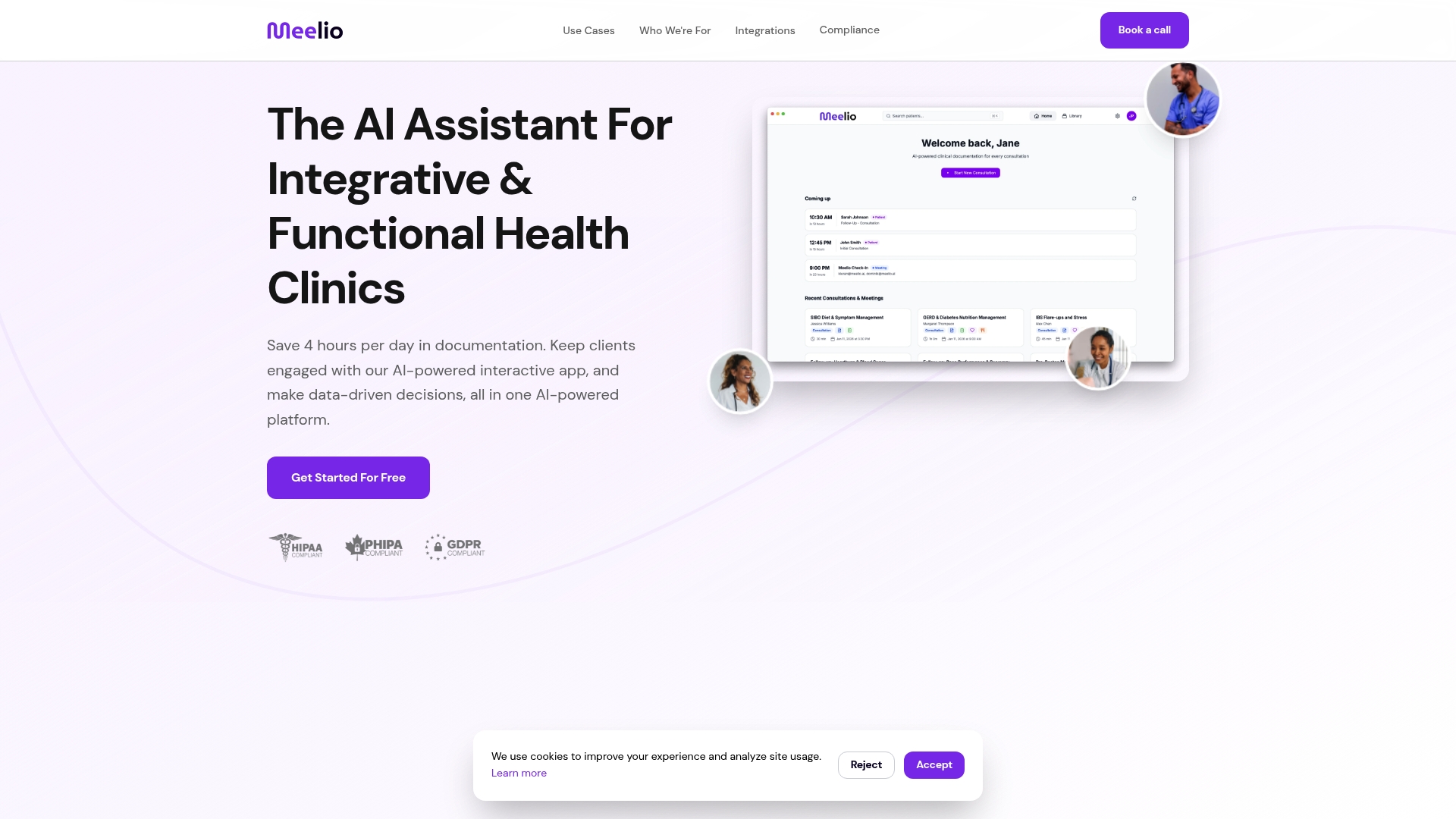
Practitioners using Meelio AI assistant for clinics save over 4 hours daily on documentation and care planning, redirecting that time toward patient interaction and clinical decision-making. The platform generates customized meal plans through AI meal planning solutions that account for food preferences, budget constraints, and therapeutic goals while maintaining nutritional adequacy. Comprehensive care plan creation via AI care planning tools ensures nothing falls through the cracks during complex multi-system interventions.
Pro Tip: Integrate AI workflow tools early in your personalization journey rather than waiting until manual processes become overwhelming. The learning curve pays dividends through improved patient outcomes, enhanced practice efficiency, and reduced clinician burnout.
What types of data are essential for personalizing nutrition?
Comprehensive personalization requires genetic profiles revealing nutrient metabolism variants, microbiome composition showing gut health status, hormonal panels assessing metabolic and reproductive function, and lifestyle tracking capturing food-mood patterns. Continuous glucose monitors provide real-time metabolic data impossible to obtain through static lab testing. Multi-modal data integration improves dietary precision by revealing how different biological systems interact to shape individual food responses and nutrient needs.
How can AI improve personalized nutrition in clinical practice?
AI processes multi-omics datasets including genomics, metabolomics, and microbiome composition alongside lifestyle factors to generate tailored dietary recommendations. Machine learning algorithms identify patterns predicting individual responses to specific foods, macronutrient ratios, and meal timing strategies. This enhances efficiency by automating data synthesis that would take hours manually while improving patient adherence through recommendations aligned with their unique biology and preferences.
What are common limitations of personalized nutrition approaches?
High costs for comprehensive testing and AI platforms create accessibility barriers limiting patient reach and raising equity concerns. Genetic testing often contributes small effect sizes compared to behavior change, leading to overhyped expectations about DNA-based personalization. Cost-effectiveness analysis remains limited, and many practitioners lack training in interpreting complex multi-omics data. Success depends more on sustainable lifestyle modification than perfect data collection, requiring balanced investment in both technology and coaching support.
How do functional medicine protocols support personalized nutrition?
Functional medicine protocols emphasize systems biology approaches addressing interconnected root causes rather than isolated symptoms. Practitioners assess gut health, detoxification capacity, hormonal balance, and inflammatory status as integrated systems shaping nutrient metabolism. Phased interventions begin with elimination diets and foundational support before layering targeted nutraceuticals based on individual lab findings and genetic variants. This structured approach ensures comprehensive coverage while maintaining flexibility to adjust based on patient response and practical constraints.





